|
Such development would allow researchers to produce amorphous models in less than a day.Ī major hurdle in simulating systems composed of inorganic materials and biological macromolecules is to describe the strenght of the interacting forces between the inorganic surface with water and biomacromolecules. Part of our future work involves developing similar capabilities in NAMD, the premium molecular dynamics software that can run on thousand processors. In that study, the annealing steps were performed for hundreds of picoseconds using the commercial software Cerius 2 and specialized silica force fields ( glass_2.01 and glass_2.01), which are computationally expensive. The detailed protocol to obtain amorphous structures is described in a paper. The resulting structure can be refined to produce chemically-modified silica forms, by introducing hydroxyl groups or organic molecules at the exposed surface (blue atoms). Finally, the bonding connectivity is calculated using a cutoff distance of 0.2 nm. To mimic real amorphous silica surfaces, the nanodevice is annealed further, allowing the rearrangement of the surface atoms. After that, the nanodevice is sculpted by removing atoms from the amorphous cube. The initial high temperature assures the randomization of the crystalline structure. Then, the crystalline cube is annealed by increasing the temperature to 8000 K and then cooling it back slowly to 300 K. The procedure to construct an amorphous silica nanodevice is squematically pictured in the figure on the right side and briefly described in the next paragraph.įirst, a cristoballite unit cell is build and replicate it to fill a cubic volume big enough to contain the nanodevice. To obtain amorphous models, crystalline structures are randomized using a set of MD annealing steps that reproduce structural features of actual amorphous silica. However, due to the amorphous nature of silica, there are no X-ray or NMR structures that can be used as templates. 
The first compulsory step to perform molecular dynamics simulations is to specify the coordinates and connectivity of each atom of the system. For more details, take a look at the publications and links on this web site or contact the investigators listed below. In this website, we briefly describe the work that the TCBG has performed to study nanosensors based of amorphous silica. Our previous pioneering work in nanobiotechnology was focused on crystalline inorganic structures, such as gold and silicon nitride. Until recently, computer simulations of biomacromolecules and inorganic materials, such as amorphous silica and DNA, have evolved independently from each other, and joining the expertise from both areas is a formidable task. However, modeling systems that combine amorphous silica and biomolecules imposes a variety of challenges to modelers. Molecular dynamics simulations can be tailored to study those systems, becoming unique imaging tools. 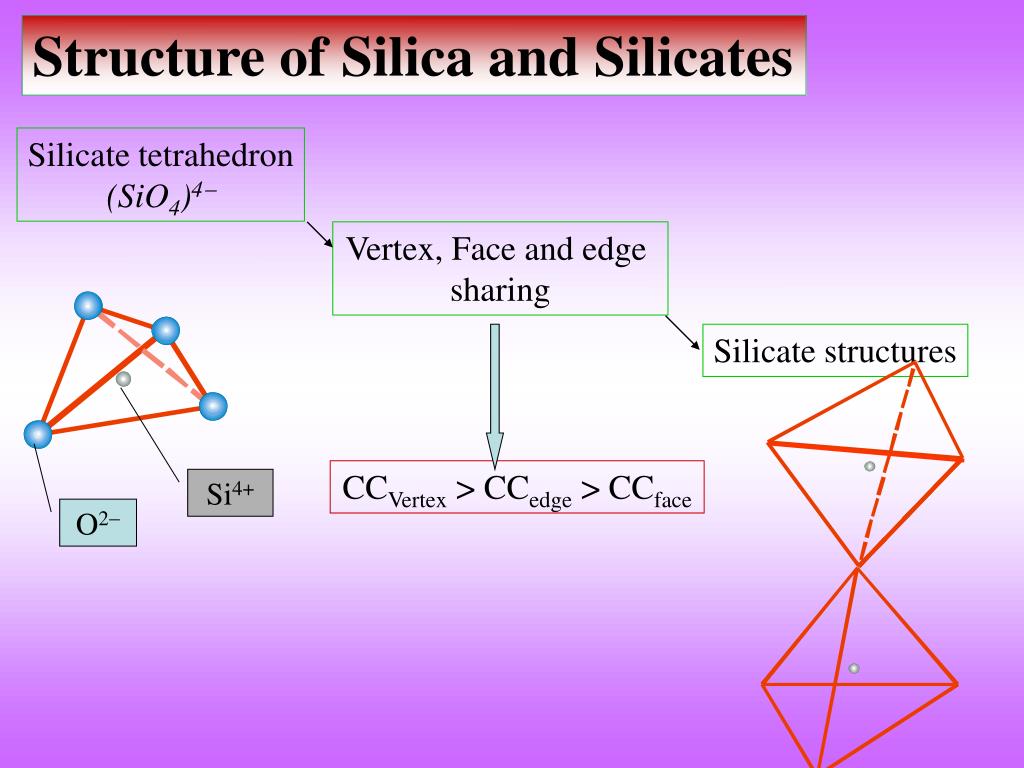
Therefore, an atomic level understanding of the interactions between biomolecules and silica is now central for further development of bionanotechnology applications.Ĭurrently, no experimental technique is yet sensitive enough to resolve atomic-scale dynamics at the amorphous interface. It has been proposed that nanopores could be used to sequence DNA with single base resolution, leading to a fast and cheap DNA sequencing technology, which promises to have a enormous impact in life sciences and personal medicine. The picture on the left side shows one of such nanodevices, a MOS nanopore manufactured on a poly-Silicon-Silica-Silicon membrane and a single-stranded DNA molecule (blue) translocating through it. In recent years, the synergy between molecular biology and nanotechnology has opened up opportunities for many applications that involve macromolecules and silica, such as nanoelectronics, self-assembly of nanostructures, microfluidics, DNA microarray technology and nanopore sensors. Furthermore, molecular biologists employ silica in resins and optical beads to study the biomacromolecules. Because of its unique properties, silica is quintessential for a broad range of applications: chips, optical fibers, and telescope glasses are manufacture on silica. Due to its mechanical resistance, high dielectric strength, and selectivity for chemical modification, amorphous silica has also become a key material in microelectronics and chromatography. Amorphous silica (SiO2) is an inorganic material commonly used in semiconductor circuits to isolate different conducting regions.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
